NIOS Class 10 Science Chapter 26 Question Answers
INTEXT QUESTIONS 26.1
1. A chemical substance may occur as an element, mixture, or compound. To which category does air belong?
Ans: Mixture
2. Name the major constituents of air. Which constituents are inevitable for survival of plants and animals?
Ans: Nitrogen and oxygen; oxygen
3. If you were to compare the relative amounts of nitrogen and oxygen in the atmosphere. Which will be four times the other?
Ans: Nitrogen
4. Air also contains water vapour. But is its percentage in air the same at all Places?
Ans: It varies from place to place.
INTEXT QUESTIONS 26.2
1. Why is oxygen essential for life? What would happen if there is no oxygen in air?
Ans: Needed for respiration by plants and animals; animals would die.
2. Carbon dioxide acts as food for plants. Name the process in which it is utilized for making food.
Ans: Photosynthesis
3. What is dry ice and what is it used for?
Ans: Solid CO2, used as a refrigerant
4. If you were to analyse all proteins, you would find a particular element common to all. Which one is it?
Ans: Nitrogen
INTEXT QUESTIONS 26.3
1. What happens to atmospheric pressure as we climb a mountain?
Ans: It decreases with altitude
2. At high altitude, the people find their nose bleeding. Why?
Ans: It is because the blood pressure in the blood vessels of the body is much more than the air pressure at high altitude, so the capillaries burst and bleeding occurs.
3. Which layer of atmosphere is the closest to the Earth’s surface, and which is the farthest from Earth’s surface?
Ans: i) Troposphere (ii) Thermosphere
4. In which layer of atmosphere is ozone layer present?
Ans: Stratosphere
5. Name (i) a greenhouse gas, (ii) gas responsible for acid rain, (iii) chemicals causing ozone hole.
Ans: (i) Methane (ii) Sulphur oxide (SOx) (iii) Chloro fluoro carbons (CFCs)
INTEXT QUESTIONS 26.4
1. It is said that more of the Earth is water than land. How much of the Earth’s surface is covered by water?
Ans: Three-fourths
2. Name any two sources of water.
Ans: Rain and sea (or any other)
3. Is rainwater pure or impure? Give one reason to support your answer.
Ans: Pure/ Distilled
4. What does chlorination do to water in order to purify it?
Ans: Kill microorganisms
5. I could not form lather with soap while washing my hands. Which type of water was it?
Ans: Hard water
6. Name the type of hardness caused to water due to the presence of bicarbonates of Ca2+ or Mg2+.
Ans: Temporary hardness
7. Name the type of hardness caused due to the presence of chloride or sulphate of Ca2+ or Mg2+.
Ans: Permanent hardness
8. Which type of hardness is removed by the following:
(i) boiling _____________
(ii) ion exchange method. ______________
Ans: (i) Temporary, (ii) permanent
9. Is water a polar or a nonpolar solvent? Why do you think so?
Ans: Polar—reasons to be given
10. At what temperature does water take up least space?
Ans: 40°C
INTEXT QUESTIONS 26.5
1. State any two uses of harvesting rainwater?
Ans:
- i. It conserves valuable groundwater.
- ii. It reduces local flooding and drainage problems,
- iii. It decreases landscaping and property maintenance needs
- iv. It provides quality water for many household needs;
- v. It can be used for domestic purposes (Any two)
2. How would industries and agriculture suffer in the event of acute scarcity of water?
Ans:
- Industries: coolant purpose, production of steam, and use as a solvent for many chemicals would be affected.
- Agriculture: irrigation of crops, germination of seeds, and growth of plants would be affected.
3. What does rainwater do to groundwater?
Ans: Raises the declined water level.
4. Why does rainwater prove to be suitable for washing with soap?
Ans: Because rainwater is in the form of soft water.
TERMINAL EXERCISES
1. Multiple-choice type questions.
i. Air is
a) compound
b) element
c) mixture
d) none of these
Ans: c) mixture
ii. Major components of air are
a) CO2 and H2O
b) N2 and O2
c) CO2 and He
d) H2O and Xe
Ans: b) N2 and O2
iii. The instrument used to measure humidity is
a) barometer
b) hygrometer
c) lactometer
d) thermometer
Ans: b) hygrometer
iv. Water has maximum density at
a) 0°C
b) 10 °C
c) 5°C
d) 4 °C
Ans: d) 4 °C
2. List the utility of oxygen and nitrogen in our lives.
Ans:
- Oxygen
- Necessary for respiration.
- Supports combustion of materials.
- Supports aquatic life by dissolving in water.
- A mixture of oxygen and nitrogen is used as anesthesia in surgical operations.
- Nitrogen
- Nitrogen is the main constituent of proteins.
- Several amino acids that contain nitrogen join together to form a protein, the building block of the body.
- Nitrogen subdues the activity of oxygen. If the concentration of oxygen in air is increased, processes like metabolism, combustion, and corrosion are sped up, and this shall have a harmful effect.
3. What is atmospheric pressure?
Ans: The force of the air column acting per unit area of a surface results in a pressure exerted by the atmosphere. This pressure is called atmospheric pressure.
4. How does the atmospheric pressure depend on altitude?
Ans: The atmosphere is much denser near the surface of Earth than at higher altitudes. In fact, the density of air decreases very rapidly with increasing distance from Earth. Therefore, atmospheric pressure also decreases with altitude.
5. Give an activity which proves that air exerts pressure.
Ans:
- Aim: To show that air exerts pressure
- Requirement: An empty polythene bottle of mineral water and some hot water.
- Procedure
- Take an empty bottle of mineral water.
- Take some hot water in it and tightly screw its cap in order to make it airtight.
- Pour cold water on the bottle.
- Observation: You will find that the bottle collapses and becomes misshapen when the water vapor inside cools and condenses into water.
6. What is relative humidity?
Ans: Relative humidity is the ratio of the mass of water vapour actually present in a certain volume of air at room temperature to the mass of water vapour required to saturate the same volume of air at that temperature.
7. What is the different sources of water? Mention any two.
Ans: The natural sources of water are rain, springs, wells, rivers, and seas.
- (a) Rainwater: Rainwater is considered to be the purest form of natural water (distilled water), free from impurities. Water from the sea and rivers evaporates into water vapour by the heat of the sun. During this process of evaporation, impurities are left behind. When the water vapours go high up in the air, it condenses to form clouds. The water drops come down as rain.
- (b) Spring water: Springs are formed by percolation of rainwater into soil. Springs supply water to wells and lakes.
- (c) Well water: The rainwater seeps through the soil and goes down and is stored over rocks or a hard earth crust. On digging the well, this underground water becomes available to us.
- (d) River water: Rivers are formed by the melting of snow on the mountain, and also sometimes from rainwater.
- (e) Sea water: Out of all the sources, sea water is the largest natural source of water. However, it is also the source of common salt and other important chemicals.
8. Why is water called as universal solvent?
Ans: Water has a unique property of dissolving a large number of substances, from solids
such as common salt, sugar to gases like oxygen, carbon dioxide, etc. As so many substances dissolve in water, it is called a universal solvent.
9. What are the different ways to purify drinking water? What is the role of chlorination?
Ans: Purification of water to make it suitable for drinking
- Decantation is the process of separation of solids from the liquid by allowing the former to settle down and pouring off the latter. Water is kept in a vessel for some time to allow suspended insoluble impurities to settle down. Clean water can now be carefully poured into another clean vessel without disturbing the settled impurities that are left behind. However, this water must be further treated to make it suitable for drinking.
- By filtration, the insoluble impurities can be removed. It is a more effective method than decantation to remove finer particles of insoluble impurities. A piece of clean and very fine cloth can be used as a cheap and easily available filter. When water is poured through it, the filter stops the insoluble impurities, and clean water passes through it.
- By chlorine treatment, small living organisms and bacteria are killed. If required, treated water may be filtered to remove insoluble impurities.
10. What do you mean by hard and soft water? Explain the types of hardness in water.
Ans: Water forms lather with soap, which is used for cleaning purposes. It is called soft water. Sometimes water from some sources, like rivers or hand pumps, does not produce any lather with soap. It is called hard water. The hardness of water is due to the presence of salts of magnesium and calcium in water.
11. How are the temporary and permanent hardness removed from water?
Ans:
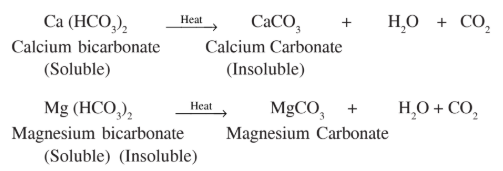
- a) Temporary hardness: Temporary hardness of water is due to the presence of soluble bicarbonates of calcium and magnesium. It is also called carbonate hardness. It can be removed by boiling and by the soda lime process.
- (i) By boiling: Upon boiling hard water, calcium or magnesium bicarbonate present in it are decomposed to give magnesium or calcium carbonate. These carbonate salts are insoluble in water. They settle down easily, and water can be decanted.
- (ii) By soda lime process (Clark’s method): When a calculated amount of lime is added to hard water, then the soluble bicarbonates are converted to insoluble carbonates.
- b) Permanent hardness: Permanent hardness of water is due to the presence of soluble chlorides and sulphates of calcium and magnesium. It is also known as non-carbonate hardness. It can be removed by the addition of washing soda or by the ion exchange method.
- (i) By addition of washing soda: The hard water is treated with the ‘calculated’ quantity of washing soda (sodium carbonate). Washing soda reacts with chloride and sulphate of calcium and magnesium to form a precipitate of calcium and magnesium carbonate. The precipitate settles down and can be removed by decantation.
- (ii) By ion. exchange method: Two types of ion exchangers can be used, namely, inorganic ion exchangers and organic ion exchangers.
- In the inorganic ion exchange process, complex compounds known as zeolites are used to soften the hard water. The salts causing hardness of water are precipitated as insoluble zeolite of calcium and magnesium, and are replaced by soluble sodium salts.
- By using an organic ion exchanger, water obtained is free from cations and anions and is known as deionized water or demineralized water.
12. Explain the following properties of water
(i) Surface tension
(ii) Density
Ans:
- (i) Surface tension: The tension exerted by molecules of water present on the surface layer is called surface tension. Due to this tension, water drops try to occupy a minimum surface area. Hence, water droplets always tend to take the shape of a sphere.
- (ii) Density: As the temperature rises from 0°C to 4°C, it actually contracts. However, from 4°C upwards it expands like any other liquid. This means that water takes up the least space at 4°C. It has the highest density at this temperature and will sink through warmer or colder water around it.
13. What is rainwater harvesting? How is it beneficial for everyday life?
Ans: Rainwater harvesting means collecting rainwater on the roofs of buildings and storing it underground. Not only does this recharge arrest the underground depletion of water, but it also raises the declining water level. The benefits of harvesting rainwater can be summarised as follows.
- Conserves valuable groundwater.
- Reduces local flooding and drainage problems.
- Decreases landscaping and property maintenance needs.
- Provides excellent quality water for many household uses.
- It can be used for domestic purposes such as growing vegetables, flowers, trees and shrubs, and seedlings in a greenhouse, etc.
14. Why is the presence of carbon dioxide in atmosphere essential? Give two reasons.
Ans:
- Carbon dioxide is required for photosynthesis in plants, which will generate oxygen for all living beings. During photosynthesis, plants absorb carbon dioxide and water vapour from the atmosphere and convert them into carbohydrates (Sugars) in the presence of chlorophyll and sunlight.
- Carbon dioxide dissolves in water to form carbonic acid, H2CO3, which reacts with rocks that contain calcium carbonate (CaCO3) or magnesium carbonate (MgCO3 form Ca(HCO3)2 and Mg(HCO3)2 salts. These salts give the taste of natural water and also supply Ca2+ and Mg2+ ions to the plants, which are necessary for their growth.
15. Give any two medical uses of oxygen.
Ans:
- Oxygen is given to patients suffering from asthma or gas poisoning, and for artificial respiration in hospitals.
- A mixture of oxygen and nitrous oxide is used as anesthesia in surgical operations.
16. What are primary and secondary air pollutants? Give one example of each.
Ans:
- Primary pollutants, which are directly emitted into the atmosphere, such as carbon monoxide from the exhaust of a motor vehicle. Eg., carbon monoxide, carbon dioxide, sulphur oxides, nitrogen oxides, etc.
- Secondary pollutants, which are not emitted directly into the atmosphere but are formed in the air when primary pollutants interact. Eg., Photochemical smog, ground-level ozone, etc.
17. What are the sources of the following pollutants : (i) Chlorofluorocarbons, (ii) Nitrogen oxides, (iii) Particulate matter
Ans:
- (i) Chlorofluorocarbons: Refrigerators and air conditioners.
- (ii) Nitrogen oxides: Lightning and fuel combustion in vehicles.
- (iii) Particulate matter: Volcanoes, dust storms, and burning of fuels.
18. Why are the following substances considered air pollutants: (i) Carbon monoxide, (ii) Carbon dioxide, (iii) Sulphur oxides, (iv) Volatile organic compounds
Ans:
- (i) Carbon monoxide: Poisonous to humans.
- (ii) Carbon dioxide: Reduces oxygen levels and causes the greenhouse effect.
- (iii) Sulphur oxides: Cause acid rain when mixed with rainwater in the atmosphere.
- (iv) Volatile organic compounds: They are greenhouse gases and suspected carcinogens.
19. What are (i) photochemical smog and (ii) ground-level ozone?
Ans:
- (i) Photochemical smog: Photochemical smog (smoke + fog) formed by the action of ultraviolet light from the sun on particulate matter or formed due to the burning of coal and petrol in an atmosphere containing SO2. It prevents the dissipation of pollutants and causes breathing disorders.
- (ii) Ground-level ozone: Ground-level ozone (O3) is formed from NOx and VOCs. It is a constituent of smog. Normally, ozone occurs in the stratosphere and prevents UV radiation from reaching Earth’s surface. At ground level, when inhaled, it is harmful to the health of humans and animals.
20. What are (i) point source pollution and (ii) non-point source pollution? Give one example of each.
Ans:
- Point source pollution refers to contaminants that enter a water body from a single identifiable source, such as a pipe or a ditch.
- Non-point source pollution refers to diffused contamination that does not originate from a single discrete source but is the cumulative effect of contaminants gathered from a large area, such as the leaching of fertilisers and pesticides from agricultural land.
21. Dive into any two of water for each of the following purposes (i) domestic, (ii) industrial, and (iii) agricultural
Ans:
(i) Domestic
- It is used for cooking food, washing utensils and clothes, and cleaning the floor of houses.
- It is also used for whitewashing.
- It is used to take a bath.
- Water provides a good medium for extracting the body waste, such as urine, stool, or perspiration. The salts and the nutrients of the food dissolve in water. Therefore, these nutrients are easily
- absorbed by our body.
- Water helps in the assimilation of many nutrients present in food. Please recall the role of water as a universal solvent.
(ii) Industrial
- Water is used as a coolant in automobiles as well as in industries.
- It is also used in the production of ice.
- It is used for the production of steam in industrial boilers and in steam engines.
- It is used as a solvent in many industrial processes.
- Water is used to prepare many chemical compounds. For example, H2SO4 is prepared by dissolving SO3 in water, and HNO3 is prepared by dissolving NO2 in water.
- Water is also used to prepare fuels like hydrogen gas and water gas.
(iii) Agricultural
- In the agriculture sector, water is used for the irrigation of crops.
- It helps in the germination of seeds and the growth of plants.
- The nutrients provided by fertilizers to the soil are soluble in water. These dissolved nutrients/fertilizers are easily absorbed by the plants.
- Water is also required (along with carbon dioxide) for the preparation of food by plants (photosynthesis).
- It also acts as a medium for the transportation of nutrients and minerals from one part of the plant to another.
- Water provides a home for aquatic plants and animals.
22. What do you mean by conservation of water? How is it useful?
Ans: Water conservation is the collection of water by making dams and reservoirs, creating groundwater structures such as wells, recycling of used water, and desalination of water. Recharging of groundwater has become necessary, which is being done through rainwater harvesting.
- Harvesting rainwater not only helps reduce the possibility of flooding, but it also decreases the community’s dependence on groundwater for domestic uses.
- Rainwater is perfectly suited for landscape irrigation, growing vegetables and flowers, use in room coolers, washing, and many other applications.
- Being soft water, rainwater is used for washing purposes as it prevents hardness deposits, and there is no problem of soap scum.
- Harvested water may also be used for personal consumption, but it must be filtered and treated prior to use.
- By reducing runoff of the rainwater that falls on your house or field, you can put a valuable water resource to work around your house.
23. You are in charge of the residents’ welfare association. Prepare two slogans to make residents aware of need for conservation of water.
Ans: Write in your own words.
24. The atmospheric envelope around living beings is the provider of the gases essential for their survival. Which are these gases, what is their proportion in air, and how are they responsible for survival?
Ans: Oxygen and carbon dioxide are the essential gases.
- All living beings require oxygen to sustain life. It constitutes 20.09% of the volume in air.
- Carbon dioxide is essential for plants to perform photosynthesis, which eventually provides food for all living beings. It is just 0.033% in the air.
25. How does rainwater pollute water bodies with air pollutants?
Ans: Rainwater pollutes water bodies with air pollutants due to acid rain.
- Oxidation of sulphur dioxide (SO2) to sulphur trioxide (SO3) results in the formation of sulphuric acid (H2SO4), which causes acid rain.
- Nitrogen oxides (NOx), especially nitrogen dioxide, NO2, catalyse the oxidation of SO2 to SO3 and indirectly cause acid rain.
26. Recall five properties of water and write a paragraph justifying that water is an indispensable resource.
Ans:
- Universal Solvent: It has a unique property of dissolving a large number of substances, starting from solids such as common salt, sugar, to gases like oxygen, carbon dioxid,e etc. Indeed, as so many substances dissolve in water, it is called a universal solvent. This property of water is useful for plants to take their food materials and minerals from the soil. It helps us to absorb the food that we eat.
- Polar nature: Water is a very effective solvent for ionic compounds. Although water is an electrically neutral molecule, it has a small positive charge (on the H atoms) and a negative charge (on the O atom), Therefore, it is polar in nature and can dissolve ionic compounds.
- Surface Tension: Due to this tension, water drops try to occupy a minimum surface area. Hence, water droplets always tend to take the shape of a sphere.
- Capillary Rise: This property of the rise of water inside a capillary is called capillarity or capillary action. This is the property by which water from the soil enters the leaves and branches of the plants through the stems.
- Density: Water behaves in an unusual way when it is heated from 0°C. As the temperature rises from 0°C to 4°C, it actually contracts. However, from 4°C upwards it expands like any other liquid. This means that water takes up the least space at 4°C. It has the highest density at this temperature and will sink through warmer or colder water around it. The density of water at 4°c is 1g/m³.
27. How does boiling of hard water convert it such that it can be used for washing clothes?
Ans: Upon boiling hard water, calcium or magnesium bicarbonate present in it are decomposed to give magnesium or calcium carbonate. These carbonate salts are insoluble in water. They settle down easily, and water can be decanted.
Additional Study Materials
- Chapter 19 Classification of Living Organisms
- Chapter 20 History of Life on Earth
- Chapter 21 Building Blocks of Life – Cell and Tissue
- Chapter 22 Life Process I: Nutrition, Transportation, Respiration, and Excretion
- Chapter 23 Life Processes II: Control and Coordination
- Chapter 24 Life Processes III: Reproduction
- Chapter 25 Heredity




