NIOS Class 10 Science Chapter 8 Question Answers
INTEXT QUESTION 8.1
1. Put the following substances in acid or base bottle.
(a) Milk of magnesia
(b) gastric juice in humans
(c) soft drinks
(d) lime water
(e) vinegar
(f) soap
Ans:
- Acidic : (b), (c) and (e)
- Basic : (a), (d) and (f)
2. What will happen if you add a drop of the following on a cut unripe apple, curd, caustic soda solution and soap solution?
(i) phenolphthalein
(ii) litmus
Ans:
- Phenolphthalein: Colourless on unripe apple and pink in solutions of caustic soda and soap.
- Litmus: Red on unripe apple and curd, and blue in solutions of caustic soda and soap solution.
INTEXT QUESTIONS 8.2
1. Name the substances in which the following acids are present:
(a) Ethanoic acid (b) Tartaric acid
Ans: (a) Vinegar (b) tamarind
2. Which of these acids would be partially dissociated in their aqueous solution?
(a) HBr (b) HCN
(c) HNO3 (d) C2H5COOH
Ans: (b) and (d)
3. An acid reacts with a substance X with liberation of a gas, which burns with a ‘pop’ sound when a burning matchstick is brought near it. What is the nature of X?
Ans: It must be a metal.
4. An acid reacts with a substance Z with the liberation of CO2 gas. What can be the nature of Z?
Ans: It may be either a metal carbonate or a hydrogen carbonate.
5. Which of the following oxides will react with a base?
(a) CaO (b) SO2
Ans: SO2
INTEXT QUESTIONS 8.3
1. Why does the colour of dry blue litmus paper remain unchanged even when it is brought in contact with HCl gas?
Ans: It is because HCl gas does not contain H+(aq) ions and is non-acidic
2. How does water help in dissociation of acids and bases?
Ans:
- (i) The heat released in the dissolution process helps in the dissociation process by overcoming the forces that hold the hydrogen atom or the hydroxyl group in the molecules of the acid or the base, or in breaking the chemical bond holding them.
- (ii) Presence of water weakens the electrostatic forces between anions and cations
3. Identify the nature of the following aqueous solutions (whether acidic, basic or neutral)
(a) Solution A: [H+] < [OH–]
(b) Solution B: [H+] > [OH–]
(c) Solution C: [H+] = [OH–]
Ans:
- (a) Solution A – basic
- (b) Solution B – acidic
- (c) Solution C – neutral
INTEXT QUESTIONS 8.4
1. pOH of a solution is 5.2. What is its pH? Comment on the nature (acidic, basic or neutral) of this solution.
Ans:
Since pH + pOH = 14
pH = 14 – pOH = 14 – 5.2
= 8.8
Since pH > 7.0, it is basic in nature
2. pH of a solution is 9. What is the concentration of H+ ions in it?
Ans:
pH = –log[H+] = 9
∴ log[H+] = –9
or [H+] = 10–9 mol L–1
3. What is the nature (whether acidic, basic or neutral) of the following solutions?
(a) Solution A: pH = pOH
(b) Solution B: pH > pOH
(c) Solution C: pH < pOH
Ans:
(a) Solution A — neutral
(b) Solution B — basic (since [H+] < [OH–] in it)
(c) Solution C — acidic (since [H+] > [OH–] in it)
INTEX QUESTIONS 8.5
1. Identify acid radical and basic radical in CaSO4.
Ans:
Acid radical SO42–
Basic radical Ca2+
2. CuSO4 was prepared by reacting an acid and a base. Identify the acid and the base that must have been used in this reaction.
Ans:
Acid: H2SO4 (corresponding to the acid radical SO42–)
Base: Cu(OH)2 (corresponding to the basic radical Cu2+)
3. Which one of the following is the correct formula of plaster of Paris?
CaSO4.H2O or 2CaSO4.H2O
Ans: 2CaSO4.H2O
TERMINAL EXERCISE
A. Objective Type Questions
I. Mark the correct choice
1. Lemon juice contains
(a) tartaric acid
(b) ascorbic acid
(c) acetic acid
(d) lactic acid
Ans: (b) ascorbic acid
2. Aqueous solutions of acids conduct electricity. This shows that
(a) They contain H+ ions
(b) They contain OH– ion
(c) They contain cations and anions
(d) They contain both H+ and OH– ions
Ans: (a) They contain H+ ions
3. Which of the following is not a strong acid?
(a) HCl
(b) HBr
(c) HI
(d) HF
Ans: (d) HF
4. Self-dissociation of water produces
(a) a large number of H+ ions
(b) a large number of OH– ions
(c) H+ and OH– ions in equal numbers
(d) H+ and OH– ions in unequal numbers
Ans:
5. In any aqueous basic solution
(a) [H+] > [OH–]
(b) [H+] < [OH–]
(c) [H+] = [OH–]
(d) [H+] = 0
Ans: (c) [H+] = [OH–]
6. In an aqueous solution of HCl which of the following species is not present?
(a) H+
(b) OH–
(c) HCl
(d) Cl–
Ans: (b) OH–
7. Which of the following is not a raw material for manufacturing washing soda?
(a) Lime stone
(b) Ammonia
(c) Slaked lime
(d) Sodium chloride
Ans: (c) Slaked lime
II. Mark the following statements as true (T) or false (F):
1. Acids furnish H+ ions only in the presence of water.
Ans: True
2. Lime water turns blue litmus red.
Ans: False
3. HF is a strong acid.
Ans: False
4. H2 gas is produced when acids react with metal oxides.
Ans: False
5. Corrosive action of acids is due to H+ ions present in them.
Ans: True
6. When the pH of the rain water become more than 5.6 it is called acid rain.
Ans: False
7. Aqueous solutions of all the salts are neutral in nature i.e. neither acidic nor basic in nature.
Ans: False
III. Fill in the blanks
1. Acids taste …………………. while bases taste ………………….
Ans: Sour and bitter
2. Milk of magnesia turns …………………. litmus ………………….
Ans: Red, blue
3. One mole of sulphuric acid would furnish ……… mole/s of H+ ions and …….. moles of SO42– ions.
Ans: Two and one
4. …………………. gas is produced when acids react with metal hydrogen carbonates.
Ans: Carbon dioxide
5. Lime water turns milky on passing CO2 gas due to the formation of …………………
Ans: Calcium carbonate
6. The reaction between an acid and a base is known as ………………….
Ans: neutraliuzation.
7. Bee sting injects …………………. acid which causes severe pain and burning sensation.
Ans: formic acid
8. In NH4NO3 the acid radical is …………………. and the basic radical is …………………
Ans: nitrate (NO3-) and ammonium (NH4+)
9. Chemically baking soda is ………………….
Ans: Sodium bicarbonate
B. Descriptive Questions
1. What is an acid?
Ans: An acid is a substance which furnishes hydrogen ions (H+) when dissolved in water.
2. Give two examples of acids found in food articles.
Ans:
- Ascorbic acid (vitamin C) in lemon and many fruits
- Citric acid in oranges and lemons
3. What is a base?
Ans: A base is a substance which furnishes hydroxide ions (OH–) when dissolved in water.
4. Give two examples of bases.
Ans:
- Sodium hydroxide (NaOH) or caustic soda is used in washing soaps.
- Potassium hydroxide (KOH) or potash is used in bathing soaps.
5. What are indicators?
Ans: Substances that show one colour in an acidic medium and another colour in a basic medium are called acid-base indicators.
6. What is the colour of methyl orange indicator in (i) acidic medium and (ii) basic medium.
Ans:
- (i) acidic medium: Red
- (ii) basic medium: Yellow
7. Why do solutions of acids and bases conduct electricity?
Ans: When acids are dissolved in water, they produce ions, which help in conducting electricity. dissociation. More specifically, acids produce hydrogen ions (H+), which are responsible for all their characteristic properties. Bases produce hydroxyl ions (OH–,) which are responsible for their characteristic properties.
8. Differentiate between strong and weak acids and give one example of each.
Ans: The acids which completely dissociate in water are called strong acids. E.g., Nitric acid HNO3, HCl, etc. The acids which dissociate partially in water are called weak acids. All organic acids, like acetic aci,d and some inorganic acids are weak acids.
9. Write down the reaction between zinc and sulphuric acid.
Ans: Zinc reacts with sulphuric acid to form zinc sulphate and release hydrogen gas.
Zn + H2SO4 –→ ZnSO4 + H2 ↑
10. Which gas is evolved when an acid reacts with metal carbonates? Which other category of compounds would produce the same gas on reacting with acids?
Ans: When dilute HCl is added to sodium carbonate or sodium hydrogen carbonate, carbon dioxide gas is evolved. Metal hydrogencarbonates also release CO2 when reacting with acids.
CaCO3 + 2HCl –→ CaCl2 + H2O + CO2
11. What type of oxides react with acids? Give one examples of this type of oxide and write down the balanced equation for the reaction.
Ans: Metal oxides such as copper oxide reacts with acids. When a mixture of dilute HCl and copper oxide is mixed, the black particles of copper oxide can be seen suspended in colourless dilute hydrochloric acid.
As the reaction proceeds, the black particles slowly dissolve and the colour of the solution becomes bluish green due to the formation of copper (II) chloride (cupric chloride) – a salt.
CuO(s) + 2HCl(aq) —→ CuCl2(aq) + H2O(l)
12. What is the name given to the reaction between an acid and a base? What are the products formed in such reactions?
Ans: The reaction between an acid and a base is called neutralisation. It results in the formation of salt and water.
HCl(aq) + KOH(aq) —→ KCl(aq) + H2O(l)
H2SO4(aq) + 2NaOH(aq) —→ Na2SO4(aq) + 2H2O(l)
13. “Corrosive action of acids is not related to their strength”. Justify this statement.
Ans: Corrosive action of acids is not related to their strength. It is related to the negatively charged part of the acid. For example, hydrofluoric acid (HF )is a weak acid. Yet, it is so corrosive that it attacks and dissolves even glass. The fluoride ion attacks the silicon atom in silica glass ,while the hydrogen ion attacks the oxygen of silica (SiO2) in the glass.
SiO2 + 4HF —→ SiF4 + 2H2O
14. Give one example each of the following (i) a strong base (ii) a weak base
Ans:
- Strong base: KOH (Potassium hydroxide)
- Weak base: NH4OH (Ammonium hydroxide)
15. List three categories of substances that can react with a base. Give one example of each and write the chemical reaction involved in each case.
Ans: Bases react with
- Metals hydroxides: Zn(s) + 2NaOH(aq) ⎯→ Na2ZnO2(aq) + H2(g) ↑
- Non-metal oxides: Ca(OH)2(aq) + CO2(g) ⎯→ CaCO3(s) + H2O(l)
- Acids: HCl(aq) + KOH(aq) ⎯→ KCl(aq) + H2O(l)
16. What happens when a dry strip of each of red litmus paper and blue litmus paper is brought in contact with HCl gas? In which case a change would be observed if the strips are moistened and then brought in contact with HCl gas and what would be the change?
Ans:
If a dry strip of blue litmus paper is brought near the mouth of the test tube containing dry HCl gas, its colour does not change. When it is moistened with a drop of water and again brought near the mouth of the test tube, its colour turns red. It shows that there are no H+ ions in dry HCl gas. Only when it dissolves in water, H+ ions are formed, and it shows its acidic nature by turning the colour of the blue litmus paper to red.
17. A small palette of NaOH is kept on dry red litmus paper. Initially, no change is observed but after some time its colour starts changing to blue around the place where the palette of NaOH is kept. Explain these observations.
Ans: In dry solid NaOH, although OH– ions are present but they are not free and do not show a basic nature. On coming in contact with water, OH– ions become free and show the basic nature by changing red litmus blue.
18. How does water help in dissociation of acids and bases? Explain.
Ans: Although there are H+ ions in acids and OH– ions are present in bases, they are released only when it is dissolved in water. Thus, water helps with the dissociation of acids and bases. Thus, acidic and basic characters of different substances can be observed only when they are dissolved in water.
19. What is ‘self-dissociation of water’? Name the resulting species and give their concentrations at 25°C.
Ans: The process of dissociation of water molecules into H+ and OH- ions. The dissociation of water is extremely small, and only about two out of every billion (109) water molecules are dissociated at 25°C. As a result, the concentrations of H+(aq) and OH– (aq) ions formed are also extremely low. At 25°C (298K),
[H+] = [OH–] = 1.0 × 10–7 mol L–1
20. What is ionic product constant of water? Give its value at 25°C. Will the value change if an acid, base or a salt is dissolved in water?
Ans: In pure water as well as in all aqueous solutions at a given temperature, the product of concentrations of H+(aq) and OH–(aq) always remains constant. This product is called ‘ionic product of water’ and is given the symbol Kw. It is also called the ionic product constant of water. Thus, At 25°C (298 K), in pure water, Kw can be calculated as:
Kw = (1.0 × 10–7) × (1.0 × 10–7)
= 1.0 × 10–14
The value of the ionic product constant of water will not change if an acid, base, or salt is dissolved in water.
21. Give the relationships between the concentrations of hydrogen ions and hydroxyl ions in (i) pure water (ii) a neutral solution (iii) an acidic solution, and (iv) a basic solution.
Ans:
- (i) Pure water: In pure water H+(aq) and OH–(aq) ions are produced in equal numbers
- (ii) Neutral solution: In all neutral aqueous solutions, the concentrations of H+(aq) and OH–(aq) ions remains equal.
- (iii) Acidic solution: Acidic solution is the one in which the concentration of H+(aq) is greater than that of OH–(aq) ions.
- (iv) Basic solution: Basic solution is the one in which the concentration of H+(aq) ions is smaller than that of OH–1(aq) ions.
22. What is pH? What happens to the pH if the hydroxyl ion concentration in the solution increases?
Ans: The term pH means “power of hydrogen”. The pH is the logarithm (see box) of the reciprocal of the hydrogen ion concentration. It is written as:
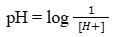
pH is the negative logarithm of the hydrogen ion concentration i.e pH = –log [H+].
When the hydroxyl ion concentration in the solution increases, the pH value increases, resulting in an alkaline solution.
23. Predict whether a given aqueous solution is acidic, basic or neutral if its pH is
(a) 7.0, (b) 11.9 and (c) 3.2.
Ans:
(a) 7.0: Neutral
(b) 11.9: Basic
(c) 3.2: Acidic
24. Calculate the pH of 1.0 ×10–4 molar solution of HNO3.
Ans: HNO3 is a strong acid.
[H+]=1.0×10−4 M
pH=−log[H+]
pH = −log(1.0×10−4) = 4
The pH = 4.
25. What is the pH of 1.0 × 10–5 molar solution of KOH?
Ans: KOH is a strong base.
[OH−] = 1.0 x 10−5 M
pOH = -log [OH−]
pOH = -log (1.0 x 10−5) = 5
pH + pOH = 14
pH = 14-5 = 9
26. What is the pH of 1.0 × 10–2 mol L–1 solution of NaCl?
Ans: NaCl is a neutral solution with a pH of 7.
27. What do you understand by the term ‘universal indicator’?
Ans: It is a mixture of a number of indicators. It shows a specific colour at a given pH. A colour guide is provided with the bottle of the indicator or the strips of paper impregnated with it, which are called pH paper strips. The test solution is tested with a drop of the universal indicator, or a drop of the test solution is put on pH paper. The colour of the solution on the pH paper is compared with the colour chart/guard, and pH is read from it.
28. What is acid rain?
Ans: When the pH of rainwater falls below 5.6, it is called acid rain.
29. What is the importance of pH for humans and animals, and our digestive system?
Ans: Most of the biochemical reactions taking place in our body are in a narrow pH range of 7.0 to 7.8. Even a small change in pH disturbs these processes.
30. Which chemical causes pain and burning sensation when somebody accidentally touches ‘nettle plant’?
Ans: Methanoic acid.
31. What is a salt? Give two examples.
Ans: Salts are ionic compounds made of a cation other than H+ ion and an anion other than OH– ion.
NaOH + HCl —→ NaCl + H2O
KOH + HNO3 —-→ KNO3 + H2O
32. How are salts obtained from an acid? Mention four types of substances that can be used for it.
Ans: Salts are obtained from an acid when reacted with metals, metal carbonates, or hydrogen carbonates. Four substances that are used to make salts are NaOH, KOH, MOH, and CaCO3.
33. Give chemical formula of (i) baking soda and (ii) washing soda.
Ans:
Baking soda: Sodium hydrogen carbonate, NaHCO3.
Washing soda: Sodium carbonate decahydrate, Na2CO3.10H2O.
34. List the raw materials required for the manufacture of baking soda and describe the process with the help of suitable chemical equations.
Ans: The raw materials required to manufacture washing soda are:
- Limestone which is calcium carbonate, CaCO3
- Sodium chloride (NaCl) in the form of brine( Conc. NaCl Solution)
- Ammonia (NH3)
Process
In Solvey’s process, carbon dioxide is obtained by heating limestone strongly,
CaCO3(s) —→ CaO(s) + CO2(g)↑
It is then passed through cold brine (a concentrated solution of NaCl in water), which has previously been saturated with ammonia,
NaCl(aq) + CO2(g) + NH3(g) + H2O(l) —→ NaHCO3(s)↓ + NH4Cl(aq)
NaHCO3 is sparingly soluble in water and crystallises out as white crystals. Its solution in water is basic in nature. It is a mild and non-corrosive base.
Action of heat: On heating, sodium hydrogen carbonate is converted into sodium carbonate and carbon dioxide is given off.
2NaHCO3 —→ Na2CO3 + H2O + CO2↑
35. Distinguish between baking powder and baking soda. Why is baking powder preferred for making cakes?
Ans: Baking powder is a mixture of sodium hydrogen carbonate and tartaric acid. Baking soda is Sodium hydrogen carbonate, NaHCO3. acid). On heating during baking, baking soda gives off carbon dioxide. It is this carbon dioxide which raises the dough. The sodium carbonate produced on heating the baking soda gives a bitter taste. Therefore, instead of using the baking soda alone, baking powder is used. The tartaric acid present in it neutralises the sodium carbonate to avoid its bitter taste. Cakes and pastries are made fluffy and soft by using baking powder.
36. Give any two uses of baking soda.
Ans:
- Used for cooking certain foods.
- It is used in medicines to neutralise the excessive acid in the stomach and provide relief.
37. What is washing soda? Give its chemical formula. How is it manufactured by Solvey’s method?
Ans: Washing soda is used for washing of clothes. It is mainly because of this chemical that the clothes washed by a washerman appear so white. Chemically, washing soda is sodium carbonate decahydrate, Na2CO3.10H2O.
Washing soda is manufactured by Solvey’s process. We have already learnt about the raw materials required and part of the process in the manufacture of baking soda. Sodium carbonate is obtained by calcination (strong heating in a furnace) of sodium hydrogen carbonate and then recrystallising from water:
2NaHCO3 —→ Na2CO3 + H2O + CO2
Na2CO3 + 10H2O —→ Na2CO3.10H2O
38. Give two uses of washing soda.
Ans:
- It is used in the manufacture of caustic soda, glass, soap powders, Borex and in the paper industry.
- For removing permanent hardness of water.
- As a cleansing agent for domestic purposes.
39. What is the chemical formula of ‘plaster of paris’? How is it manufactured? What precaution is taken during its manufacture?
Ans: 2CaSO4.H2O or CaSO4. 12H2O (calcium sulphate hemihydrate)
Raw material: Gypsum (CaSO4.2H2O) is used as the raw material.
Process: The only difference between gypsum (CaSO4.2H2O) and plaster of Paris (CaSO4.1/2H2O ) is in the amount of water of crystallisation.
When gypsum is heated at about 100° (373 K), it loses a part of its water of crystallisation to form:
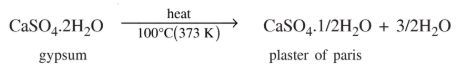
40. List any four uses of ‘plaster of paris’.
Ans:
- In making casts for the manufacture of toys and statues.
- In medicine for making plaster casts to hold fractured bones in place while they set. It is also used for making casts in dentistry.
- For making the surface of walls and ceilings smooth.
- For making decorative designs on ceilings, walls and pillars.
- For making‘ chalk’ for writing on blackboard.
- For making fire proof materials.
41. What is bleaching? Chemically, what is bleaching powder? Give its any four uses.
Ans: Bleaching is a process of removing colour from a cloth to make it whiter. Chemically, it is calcium oxychloride, CaOCl2. Its uses are,
- In the textile industry, for the bleaching of cotton and linen.
- In the paper industry, for the bleaching of wood pulp.
- In making wool unshrinkable.
- Used as disinfectant and germicide for the sterilisation of water.
- For the manufacture of chloroform.
- Used as an oxidising agent in the chemical industry.
42. List the raw materials required and the method of manufacture of bleaching powder. Write the equation for the reaction involved.
Ans: The raw materials required for the manufacture of bleaching powder are:
Slaked lime, Ca(OH)2 Chlorine gas, Cl2
Additional Study Materials
- Chapter 1 Measurement in Science and Technology
- Chapter 2 Matter in our Surroundings
- Chapter 3 Atoms and Molecules
- Chapter 4 Chemical Reaction and Equations
- Chapter 5 Atomic Structure
- Chapter 6 Periodic Classification of Elements
- Chapter 7 Chemical Bonding




